
Research and Development
We pursue bold science to develop medicines for the people with serious medical conditions.
We relentlessly pursue bold science to deliver on the promise of genetic discoveries and advance the future of human health.
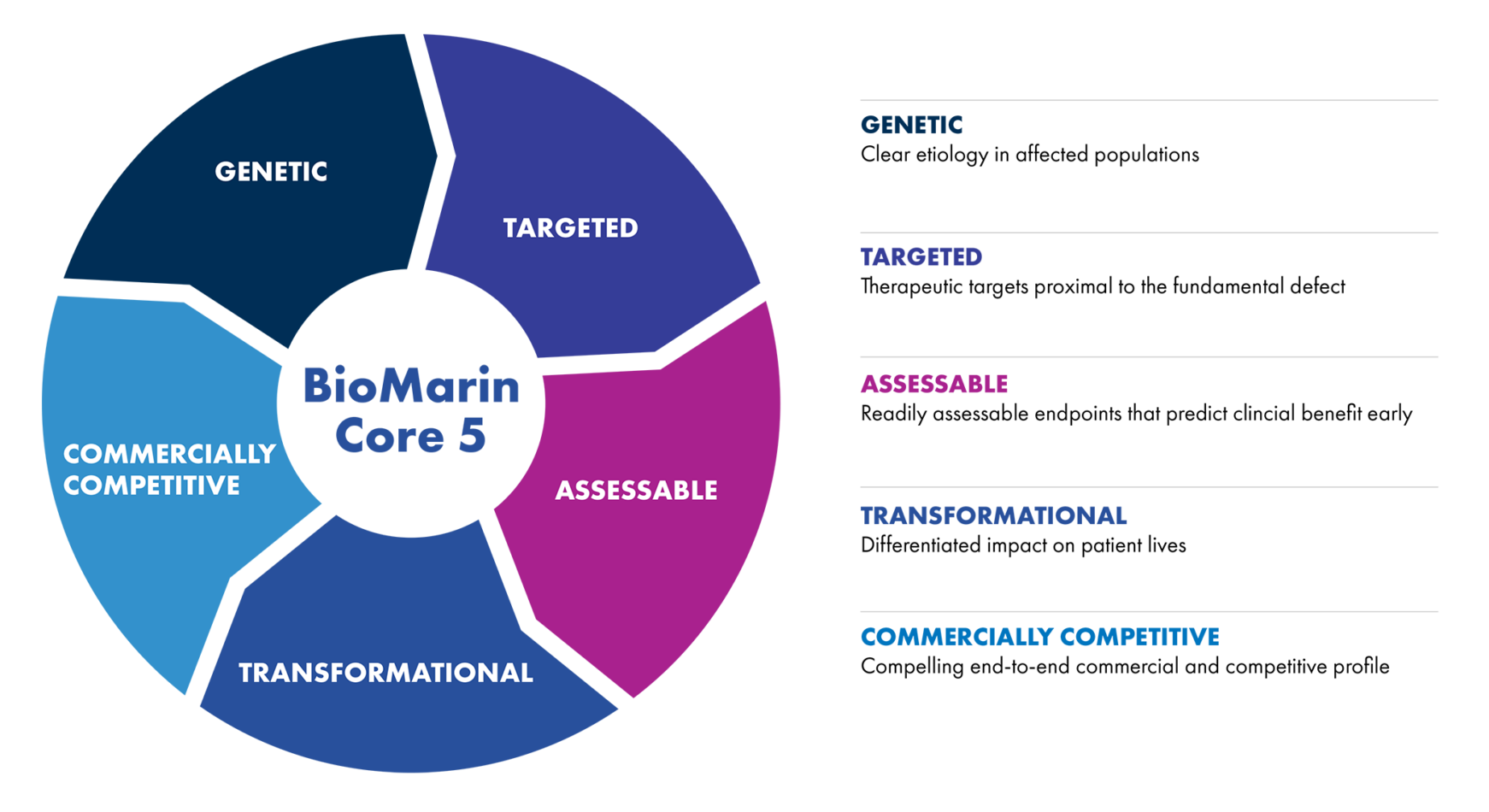
At BioMarin, we’re committed to delivering on the promise of genetic discoveries by translating them into transformative medicines. Our teams leverage their expertise in genomics and medicine to pursue breakthrough science aligned to five pillars. This has led to our unparalleled track record of introducing transformational medicines built on the foundation of genetic insights.
Our scientific foundation was built upon the research and development of enzyme replacement therapies for five rare, genetic conditions. Today, we’re leveraging deep expertise in genetics to expand our focus into new therapeutic areas to help more people living with genetic conditions around the world.
As we have looked to expand our impact, our approach to cutting-edge science has remained the same. We enlist the best of the best – people with the right technical expertise and a relentless drive to solve real problems – and create an environment that empowers our teams to pursue bold, innovative science. Our work is deeply rooted in genetic insights, meaning we target the underlying genetic changes or proximal molecular mechanisms of disease. This is the most powerful way to transform human health.
This approach has been validated again and again. It has led us to pioneering innovations for a wide range of conditions, from lysosomal storage diseases and phenylketonuria to achondroplasia and hemophilia A. We apply the same relentless spirit to the ways we create and deliver medicines. We don’t restrict ourselves to a single technology – we harness expertise in multiple treatment modalities, including biologics, gene therapy, oligonucleotides and small molecules, and apply that knowledge to pursue the greatest transformative potential for each condition.


We pursue bold science to develop medicines for the people with serious medical conditions.

Our cutting-edge facilities and comprehensive manufacturing capabilities make transformational medicines a reality.

The research we publish helps advance genetic science and foster understanding of our investigational medicines and approved products.